Leadership
- Development Core Director: Dr. Alison Gemmill, Associate Director of the HPC
- Representatives of the HPC Primary Research Areas (PRAs)
- Poverty and Inequality: Dr. Steve Morgan, Bloomberg Distinguished Professor
- Sexual and Reproductive Health: Dr. Philip Anglewicz, William G. Robertson, Jr. Professor in Population and Family Planning
- Family, Maternal and Child Health, Dr. Xiaobin Wang, Zanvyl Krieger Professor in Children’s Health
- Early-Career-Associate Representative: Dr. Dylan Jackson
Email Alison Gemmill ([email protected]), a specific PRA representative, or Dylan Jackson for inquiries regarding Development Core services.
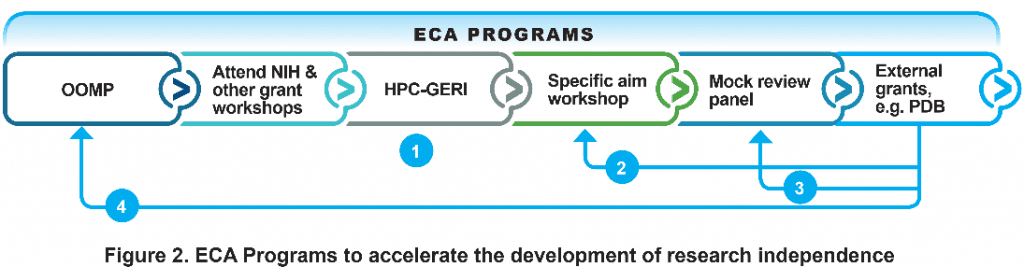
Early-Career Associate (ECA) Programs
Single-purpose grants (a total of $7,000 in a year)
- Provide funding to attend NIH grant workshop
- Support ECAs to acquire new research skills through participation short-term training programs such as summer workshops that is necessary for seeking NICHD-PDB grants
Grant writing support
- One-on-one mentorship program (OOMP)
- HPC Grant Exchange Repository Initiative (HPC-GERI)
- Specific Aims Workshop
- Mock reviews panel
- The ECA programs are subsumed under the broader development mechanisms of the Development Core as well as leveraging university resources.

Research Theme Working Groups
HPC Associates are encouraged to participate in any of these research theme working groups. Please contact the group lead for participation.
- Social Factors and Multi-Omics (Xiaobin Wang, Xiumei Hong, Pamela Surkan, Nilanjan Chatterjee, Lingxin Hao, Li Liu)
- Sensor and Health (Ciprian Crainiceanu, Lingxin Hao, Julia Burdick-Will, Marc Stein, Casey Overby Taylor)
- Reproductive Health and Family Planning (Philip Anglewicz, Linnea Zimmerman, Saifuddin Ahmed, Michele Decker, Caroline Moreau, Li Liu, and Amy Tsui)
- Innovative Demography (Li Liu, Philip Anglewicz, Andreea Creanga, Francisco Villanvicencio, Alice Gemmill, Dona Strobino, Amy Tsui, Linnea Zimmerman, Qingfeng Li, and Saifuddin Ahmed)
- Population-Scale Networks and Inequality (Angelo Mele, Lingxin Hao)
The Pilot Research Program
- Regular call (every 6 months in May and November)
- In each year, two awards (up to $20,000 each) are given to HPC Associates with priorities in senior-junior and interdisciplinary Research.
- One-time call
- One-time calls are designed to respond to special calls from NICHD-PDB for rapid proposal submissions. A past example is HPC’s Lightning Pilot Research on COVID-19.
- HPC RFAs
In our RFAs, we invite HPC Associates to submit proposals with a project length of one year. Pilot Project PIs, co-PIs, and co-investigators must be HPC Associates. The RFAs stipulate that the proposed research must represent a primary research area (PRA) leading to an NICHD-PDB application or other NICHD branches relevant to population dynamics. Associates can receive only one Pilot Award every two years. The RFAs provide step-by-step directions on the application process. The Development Core carries out targeted recruitment among ECAs and under-represented minority (URM) Associates, and identifies senior collaborators for promising but inexperienced ECAs.
- Pilot application review process
Pilot applications are first vetted by the HPC leadership to ensure priorities are given to senior-junior and interdisciplinary collaborations and the scope is within the PRAs. Once confirmed, applications are sent to three reviewers drawn from members of the Faculty Coordinating Committee, Scientific Advisory Committee, and relevant experts among HPC Associates. They are asked to score the application according to the following criteria: relevance to HPC’s PRAs, likelihood of leading to an application to PDB, ECA and URM status of the Principal and Co-Principal Investigators, whether the work is interdisciplinary, study significance, and scientific rigor. The three reviewers may also suggest revisions for and resubmission of an application. Applications receiving best scores are funded up to $20,000. Successful applicants are assigned methodological mentors through the Scientific Core. The Pilot Projects must go through IRB and obtain the human subject approval by NICHD.